CAR T Cell Therapy in India: Procedure, Cost, Side Effects And Recovery
CAR T cell therapy is a revolutionary cancer treatment that uses the body’s own immune system to fight cancer more effectively. This cutting-edge immunotherapy modifies T-cells in a laboratory to recognize and destroy cancer cells with high precision. It has shown remarkable success in treating certain blood cancers, especially when traditional treatments fail.
What is CAR T Cell Therapy?
CAR T cell therapy is an advanced cancer treatment that uses genetically modified T cells (a type of white blood cell) to identify and destroy cancer cells more effectively. It is a form of immunotherapy commonly used to treat certain blood cancers such as leukemia and lymphoma.
T-cells normally help protect the body from infections, but cancer cells can sometimes hide from them. CAR-T therapy enhances these immune cells in a lab, allowing them to better recognize and attack cancer cells.
Who is Eligible for CAR T Cell therapy?
CAR T Cell therapy is recommended for patients with certain types of blood cancers, especially when standard treatments like chemotherapy or stem cell transplant are not effective. Eligibility depends on the type of cancer, previous treatments, and overall health.
What is the cost of CAR T Cell Therapy in India?
CAR T Cell Therapy in India typically costs ₹30 lakh to ₹75 lakh, with most treatments averaging around ₹35–₹50 lakh.
How CAR T Cell Therapy Works to Destroy Cancer Cells
Diagnosis of Specific blood cancers:
CAR T cell therapy is mainly used to treat cancers such as acute lymphoblastic leukemia (ALL), diffuse large B-cell lymphoma (DLBCL), and multiple myeloma, particularly in advanced or relapsed cases.
Relapsed or refractory cancer:
Patients whose cancer has returned (relapsed) or has not responded (refractory) to treatments like chemotherapy or radiation may be suitable candidates.
Previous treatment failure:
CAR T Cell Therapy is often considered after multiple treatments, including chemotherapy, immunotherapy, or stem cell transplant, have not been successful.
Overall health and fitness:
Patients must be medically fit enough to tolerate the treatment and its side effects. Doctors evaluate organ function and general health before recommending therapy.
Targetable cancer markers:
CAR T Cell Therapy works by targeting specific proteins (antigens) on cancer cells. Patients whose cancer cells express these markers are more likely to benefit.
Clinical trial eligibility:
Some patients may receive CAR T Cell Therapy through clinical trials, providing access to newer and emerging treatment options.
CAR T Cell Therapy Procedure: Step-by-Step Treatment Process
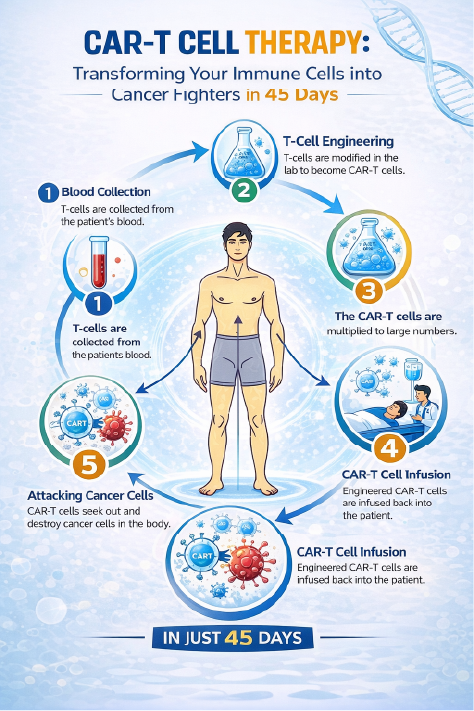
CAR T Cell Therapy is a multi-step, highly specialized process that transforms a patient’s immune cells into powerful cancer-fighting agents. Each stage is carefully designed to ensure safety and effectiveness.
1. T Cell Collection (Leukapheresis)
The process begins with collecting T cells from the patient’s blood using a procedure called leukapheresis. Blood is drawn, T Cells are separated, and the remaining blood is returned to the body.
2. Genetic Engineering of T Cells
The collected T Cells are sent to a laboratory, where they are genetically modified to produce Chimeric Antigen Receptors (CARs). These receptors help T Cells identify and attach to cancer cells.
3. CAR T Cell Expansion
The engineered T Cells are multiplied in the lab to create millions of CAR T Cells. This ensures there are enough cells to effectively target cancer when infused back into the patient.
4. Conditioning Chemotherapy
Before reinfusion, the patient receives a short course of chemotherapy. This helps suppress the existing immune system and creates space for the CAR T Cells to function effectively.
5. CAR T Cell Infusion
The modified CAR T Cells are infused back into the patient’s bloodstream through an intravenous (IV) line. This step is similar to a blood transfusion and is usually completed within a few hours.
6. Targeting and Destroying Cancer Cells
Once inside the body, CAR T Cells begin to recognize, bind to, and destroy cancer cells. These cells can continue to multiply and provide ongoing protection against cancer.
7. Monitoring and Recovery
After infusion, patients are closely monitored for side effects such as cytokine release syndrome (CRS) and neurological symptoms. Follow-up care is essential to ensure recovery and treatment success.
Types of CAR T Cell Therapy: Latest Advancements Explained
CAR T Cell Therapy has rapidly advanced over the years, with newer generations designed to improve effectiveness, safety, and long-term results. These evolving types focus on making the immune response stronger, more precise, and longer-lasting.
First-Generation CAR T Cells
The earliest version of CAR T Therapy included basic engineered T Cells that could recognize cancer cells.
- Simple structure
- Limited effectiveness
- Short-lived response
Second-Generation CAR T Cells
This is the most commonly used type today. Scientists added co-stimulatory signals to improve T Cell activity.
- Stronger immune response
- Better cancer cell targeting
- Longer-lasting effects
Third-Generation CAR T Cells
These combine two co-stimulatory domains to further enhance performance.
- More powerful T-cell activation
- Improved persistence in the body
- Enhanced tumor-killing ability
Fourth-Generation CAR T Cells (TRUCKs)
Also called “Armored CAR-T Cells”, these are engineered to release immune-boosting substances.
- Secrete cytokines to attack tumors
- Improve effectiveness in solid tumors
- Modify the tumor environment
Fifth-Generation CAR-T Cells
The latest and most advanced development in CAR-T therapy.
- Mimic natural immune system signaling
- More controlled and precise response
- Reduced side effects
Conclusion
CAR T Cell Therapy is an advanced breakthrough in cancer treatment that offers renewed hope and improved outcomes for patients with certain blood cancers. Understanding the treatment process, recovery phase, possible side effects, and cost helps patients and caregivers make well-informed healthcare decisions.When considering treatment, choosing the best hospital for CAR-T Cell Therapy in India . If you are considering CAR T Cell Therapy, it is important to consult a qualified oncologist to evaluate eligibility and receive a personalized treatment plan based on individual medical needs.
DOCTOR DETAILS:
Dr. M. Gopinathan
Consultant
Hemato-Oncology(BMT)
Frequently Asked Questions (FAQs)
No, it is a non-surgical treatment done through blood collection and infusion.
Recovery usually takes about 2 to 4 weeks, but some patients may need longer follow-up care depending on their condition and response to treatment.
CAR T cell therapy may cause Cytokine Release Syndrome (CRS), neurological side effects, infections, fever, and low blood cell counts. These complications are usually temporary and managed with close hospital monitoring and timely treatment.
Yes, it is performed under strict medical supervision with continuous monitoring.
Common side effects include fever, fatigue, flu-like symptoms, and weakness.
Contact Us
Visiting Hours
OPEN 24 hours 7 days a week.
OPD Timings : Monday to Saturday
( 9:00 AM to 5:30 PM )
Appointments
Visit the hospital
MGM Cancer Institute
No 119 & 121, Nelson Manickam Road, Raajeswari Street, Rajaram Mehta Nagar,
Aminjikarai, Chennai – 600029
At MGM Cancer Institute, we believe in curing the fear of cancer first. We understand that battling cancer is not just a physical fight, but a mental and emotional one as well. Our dedicated team is committed to providing exceptional healthcare that improves your overall well-being and eases the anxiety that comes with cancer. With a 150-bed facility in the heart of your city, we are here to support you every step of the way.
© MGM Cancer Institute. All Rights Reserved. Last updated on 20-01-2025.